Pfizer
Pfizer is a company that doesn’t need to be explained to anyone. If you don’t know somehow, they’re the maker of the covid vaccine that has the largest market share in the United States. Currently the FDA has “authorized” a monovalent version and a bivalent version, under an Emergency Use Authorization (EUA) status. The EUA status is conditional on Pfizer completing post-marketing studies and is only valid during the covid public health emergency.
Any new risks?
Lets see if they’ve added any new risks in their SEC filings.
Can you spot the difference? What did Pfizer add as a risk?
risks associated with preliminary, early stage or interim data
Now isn’t that something.
Labels, labels, labels
Let’s look at some stuff that was hidden in the PDF version of this Pfizer document.
FACT SHEET FOR HEALTHCARE PROVIDERS ADMINISTERING VACCINE (VACCINATION PROVIDERS)
EMERGENCY USE AUTHORIZATION (EUA) OF THE PFIZER-BIONTECH COVID-19 VACCINE AND THE PFIZER-BIONTECH COVID-19 VACCINE, BIVALENT (ORIGINAL AND OMICRON BA.4/BA.5) TO PREVENT CORONAVIRUS DISEASE 2019 (COVID-19).
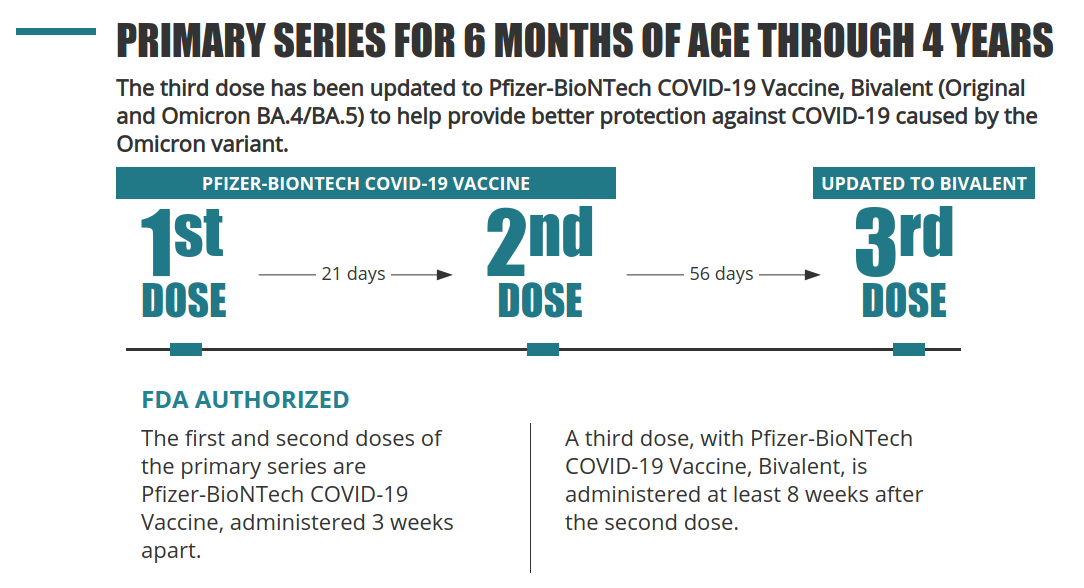
FOR 6 MONTHS THROUGH 4 YEARS OF AGE
PRIMARY SERIES
This is the fact sheet that should be given to the hospitals, clinics, and pharmacies. It gives an overview of how to administer the vaccine, has technical specifications, and answers some questions someone administering it might have.
Unfortunately, Pfizer made the PDF version not have the full side effect information. The web page did.
This is the last page on the PDF version.
I will list below a selection of what Pfizer did not include in the PDF version. The PDF version is what you get when you google it.
We won’t force it, we promise
What to even comment about this. You were never forced. Nobody forced you. There was no coercion.
We’ll make it up to you, we swear
Better submit it within a year! Sorry if you missed it!
We won’t lie about just how “authorized” it is
The possible side effects of these vaccines are still being studied. And here it’s listed as “FDA AUTHORIZED” .
Clever, AUTHORIZED. Not APPROVED.
And it does say Emergency uses of the vaccines have not been approved or licensed by FDA… on Comirnaty’s site, so it’s okay.
But the intention is clear, they are trying to confuse the status to have parents take something they believe is more well understood than it actually is.
The possible side effects of these vaccines are still being studied. This part isn’t mentioned on the Comirnaty homepage. People might want to know they’re participating in a clinical trial. Maybe it’s wrong to enroll participants like this without telling them. Just a thought.
Well, come on down, book your appointment through the drug company! It’s free!